Thrombotic risk
Restoring haemostasis in haemophilia A should be carefully balanced to avoid increasing thrombotic risk
Thrombotic events have been reported in patients treated with emicizumab1, raising concerns about such adverse events. Monitoring markers that reflect the activity of the coagulation system in patients with different treatment strategies may increase our understanding of the impact of treatment decisions on thrombotic risk in patients with inhibitors.
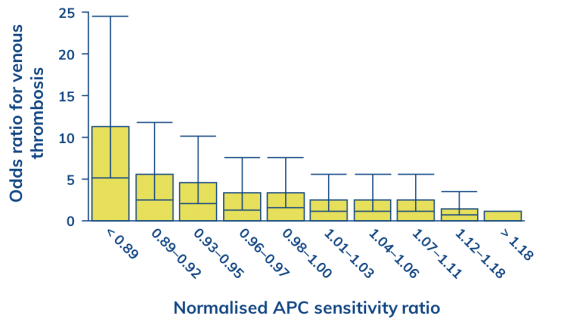
A number of plasma markers have been shown to be associated with thrombotic risk, including activated protein C (APC)2, von Willebrand factor (VWF)3 and D-dimer4. APC inactivates the activated coagulation factors VIII (FVIIIa) and V (FVa)2, a process which forms a key part of the physiological down-regulation of blood coagulation. The presence of emicizumab may impact on the APC-mediated anticoagulant pathway. Reduced overall sensitivity to APC has been shown to be associated with thrombotic risk2.
Increased risk of thrombosis is associated with a poorer response to APC
High levels of VWF have been found to be associated with increased risk of venous thrombosis3. A further established marker of venous thrombosis is the D-dimer antigen, which is generated during fibrin degradation, and is used as a standard clinical marker of venous thromboembolism4.
MOTIVATE will assess thrombotic risk with different treatment strategies by analysing the levels of these markers in treated patients.
Activated protein C (APC) levels sub-study
Sub-study lead investigators

Johannes Oldenburg
Director, Institute for Experimental Haematology and Transfusion Medicine (IHT),
Bonn, Germany

Jens Müller
Institute for Experimental Haematology and Transfusion Medicine (IHT),
Bonn, Germany
MOTIVATE will evaluate the impact of different treatment approaches on plasma levels of APC to assess thrombotic risk with different treatment strategies.
What is needed for this analysis?
Blood samples of 3 mL collected at baseline, 1 month after treatment start and every 4 months
von Willebrand factor (VWF) antigen plasma levels, VWF activity and D-dimer plasma levels sub-study
Sub-study lead investigators

Johannes Oldenburg
Director, Institute for Experimental Haematology and Transfusion Medicine (IHT),
Bonn, Germany

Jens Müller
Institute for Experimental Haematology and Transfusion Medicine (IHT),
Bonn, Germany
MOTIVATE will evaluate the impact of different treatment approaches on the risk of venous thrombosis by measuring VWF antigen plasma levels, VWF activity and D-dimer plasma levels.
What is needed for this analysis?
Blood samples of 1 mL collected at baseline, 1 month after treatment start and every 4 months
- Makris M et al. <i>J Thromb Haemost</i> 2019; 17:1269-72
- de Visser MC et al. <i>Blood</i> 1999; 93:1271-6
- Rietveld IM et al. <i>J Thromb Haemost</i> 2018; 17:99-109
- Adam SS et al. <i>Blood</i> 2009; 113:2878-87
References: